Twenty years ago, a hepatitis C diagnosis was devastating. Treatment meant months of brutal interferon injections, severe side effects, and a cure rate barely above 50%. Many patients chose not to treat at all.
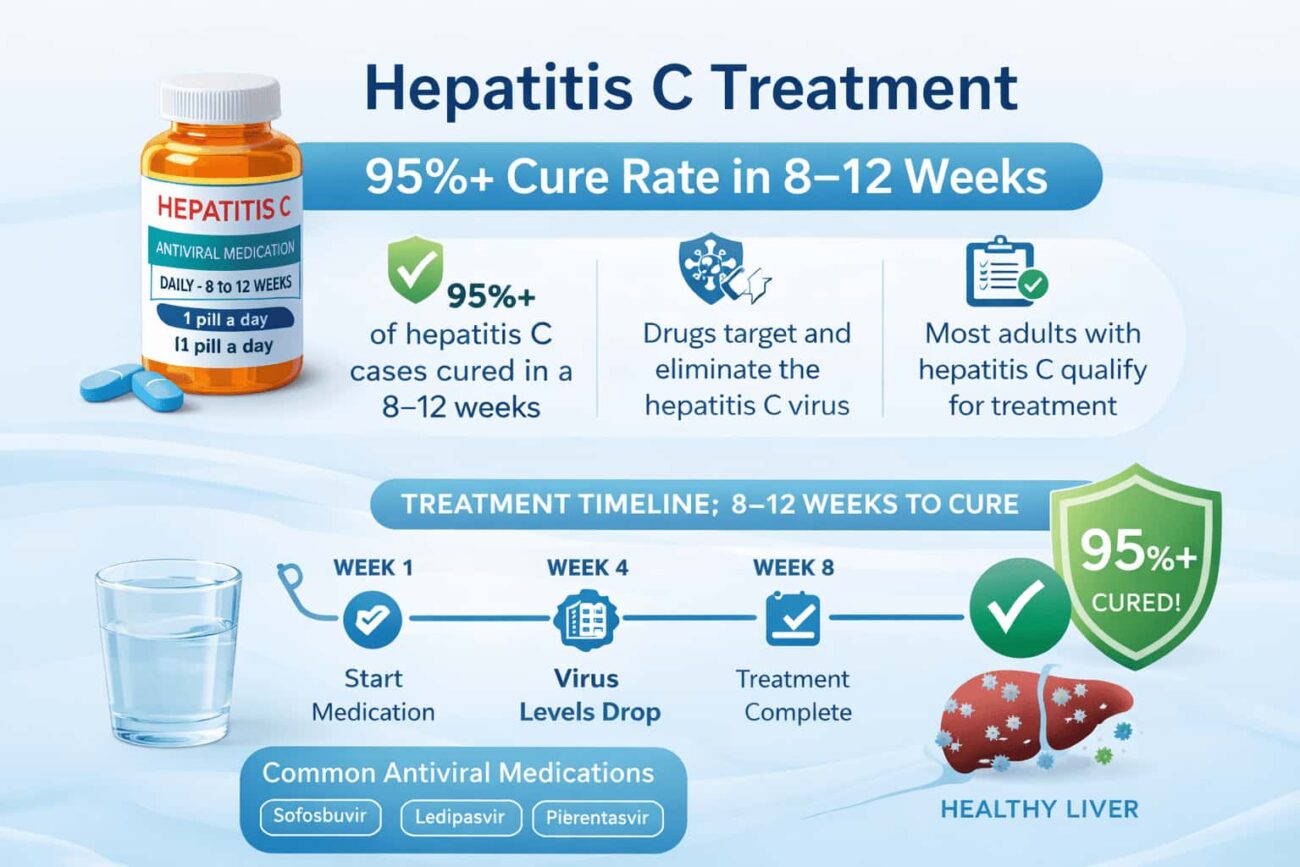
Today, the picture is completely different. Modern antiviral medication for hepatitis C — a class of drugs called direct-acting antivirals (DAAs) — cures the infection in 95–99% of patients within just 8 to 12 weeks. The drugs are taken orally, carry minimal side effects, and work across all major genotypes.
This guide explains exactly which medications are available, how they work at the molecular level, who qualifies for treatment, what to expect genotype by genotype, and the critical differences between hepatitis B and C treatment.
What Is Hepatitis C and Why Does It Need Antiviral Medication?

Hepatitis C is a blood-borne viral infection caused by the hepatitis C virus (HCV). It primarily attacks liver cells (hepatocytes), triggering inflammation that — when left untreated over years or decades — can progress to fibrosis, cirrhosis, and liver cancer (hepatocellular carcinoma).
Unlike hepatitis A or B, the body rarely clears HCV on its own. Around 75–85% of people who contract HCV will develop a chronic infection. Without treatment, approximately 15–30% of those with chronic infection will develop cirrhosis within 20 years.
This is why hepatitis C virus medication is not optional once a diagnosis is confirmed — it is the only reliable path to preventing permanent liver damage. The good news is that treatment has never been more effective, more tolerable, or more accessible.
Is Hepatitis C Curable? Here Is the Direct Answer
Yes — hepatitis C is curable. With current direct-acting antiviral (DAA) medications, 95–99% of patients achieve a sustained virologic response (SVR), which is defined as no detectable virus in the blood 12 weeks after completing treatment. SVR is considered a functional cure: the virus does not return in over 99% of cases.
When people search “hepatitis C can it be cured” or “hepatitis C is it curable,” they are often recently diagnosed and frightened. The answer is an unequivocal yes — with the caveat that treatment outcomes depend on genotype, stage of liver disease, previous treatment history, and access to medication.
What “cured” actually means in clinical terms
A cure in hepatitis C is defined as SVR12 — sustained virologic response at 12 weeks post-treatment. This means a standard PCR blood test detects zero copies of HCV RNA. Studies following SVR12 patients for 5–10 years show that fewer than 1% experience viral relapse. The liver can also partially reverse fibrosis after cure, particularly in patients without advanced cirrhosis.
Important nuance: being cured of hepatitis C does not provide immunity. A person who engages in high-risk behaviours after treatment can contract HCV again. Re-infection is possible and has been documented in populations with ongoing exposure.
How Antiviral Medication for Hepatitis C Works
The revolution in hepatitis C treatment began around 2013–2014 when the FDA approved the first generation of direct-acting antivirals (DAAs). Unlike older interferon-based regimens that worked by stimulating the immune system globally, DAAs attack specific proteins the virus needs to replicate.
HCV replication requires three key molecular targets, each addressed by a different class of DAA:
- NS3/4A protease inhibitors (suffix: “-previr”) — Block the enzyme HCV uses to process its own proteins. Without functional protease, the virus cannot produce mature copies of itself.
- NS5B polymerase inhibitors (suffix: “-buvir”) — Inhibit the RNA polymerase HCV uses to copy its genetic material. Without this enzyme, viral RNA cannot be duplicated.
- NS5A inhibitors (suffix: “-asvir”) — Disrupt the NS5A protein, which plays a critical role in viral assembly and release from liver cells.
Most modern hepatitis C medication regimens combine two or three of these drug classes in a single daily pill, achieving what is called a “pangenotypic” effect — meaning they work against multiple HCV genotypes simultaneously.
Approved Antiviral Medications for Hepatitis C (2026)
The following are the primary DAA regimens approved by the FDA and widely used globally. These represent current standard-of-care options:
| Drug / Brand Name | Drug Classes | Genotypes | Duration | Cure Rate |
|---|---|---|---|---|
| Sofosbuvir/Velpatasvir (Epclusa) |
NS5B + NS5A inhibitor | All 1–6 (pangenotypic) | 12 weeks | 95–99% |
| Glecaprevir/Pibrentasvir (Mavyret / Maviret) |
NS3/4A + NS5A inhibitor | All 1–6 (pangenotypic) | 8–16 weeks | 97–99% |
| Ledipasvir/Sofosbuvir (Harvoni) |
NS5A + NS5B inhibitor | Genotypes 1, 4, 5, 6 | 8–24 weeks | 94–99% |
| Sofosbuvir/Velpatasvir/Voxilaprevir (Vosevi) |
NS5B + NS5A + NS3/4A | All genotypes; DAA-experienced | 12 weeks | 96–98% |
| Elbasvir/Grazoprevir (Zepatier) |
NS5A + NS3/4A inhibitor | Genotypes 1, 4 | 12–16 weeks | 92–97% |
Hepatitis C Treatment by Genotype: What You Need to Know
HCV is classified into six major genotypes (1–6) and numerous subtypes. Genotype determines the genetic structure of the virus and historically influenced treatment response significantly. With the arrival of pangenotypic DAAs, genotype matters less for treatment selection — but it still affects duration, drug choice in complex cases, and response monitoring.
Genotype 1 (most common globally)
Genotype 1a and 1b account for approximately 50% of all HCV infections worldwide. Both Epclusa and Mavyret achieve SVR12 rates above 97% in treatment-naive patients. Harvoni is also commonly used for genotype 1, particularly in patients with kidney disease who need to avoid sofosbuvir-based regimens.
Genotype 2
Genotype 2 historically responded well to older interferon-based treatment and responds equally well to modern DAAs. Epclusa achieves 99% SVR12 in treatment-naive genotype 2 patients with 12 weeks of therapy.
Hepatitis C Treatment Genotype 3 — The Most Challenging
Genotype 3 is the most clinically challenging to treat, particularly in patients with advanced fibrosis or cirrhosis. It is prevalent across South Asia (India, Pakistan, Bangladesh) and parts of Southeast Asia.
Standard Epclusa achieves around 89–95% SVR12 in genotype 3 patients without cirrhosis, but this drops in cirrhotic patients — especially those who have previously failed treatment. For these cases, Vosevi (sofosbuvir/velpatasvir/voxilaprevir) is the preferred salvage regimen, achieving SVR12 rates above 95% even in previously-treated cirrhotic genotype 3 patients.
Mavyret (glecaprevir/pibrentasvir) for 12–16 weeks is also an effective genotype 3 option, particularly for treatment-naive patients, with SVR12 rates of 95–98%.
Genotypes 4, 5, and 6
These genotypes are less common globally but prevalent in specific regions — genotype 4 in the Middle East and Egypt, genotype 6 across East and Southeast Asia. Both pangenotypic regimens (Epclusa and Mavyret) achieve high SVR12 rates (95–99%) for these genotypes with standard 12-week courses.
Recommended first-line treatment by genotype:
- Genotype 1: Epclusa or Mavyret (8–12 weeks)
- Genotype 2: Epclusa or Mavyret (8–12 weeks)
- Genotype 3 (no cirrhosis): Mavyret 12 weeks or Epclusa 12 weeks
- Genotype 3 (with cirrhosis or prior treatment failure): Vosevi 12 weeks
- Genotype 4: Epclusa, Mavyret, or Zepatier (12 weeks)
- Genotypes 5 & 6: Epclusa or Mavyret (12 weeks)
New Hepatitis C Treatment Options in 2025–2026
The DAA era began around 2013, but research has continued to refine regimens. The most significant new hepatitis C treatment developments in recent years include:
- Shorter treatment durations — Treatment-naive patients without cirrhosis can now often be cured in just 8 weeks with Mavyret, down from the 24–48-week interferon courses of a decade ago.
- Retreatment regimens for DAA failures — Vosevi (triple therapy) was specifically developed for patients who failed an earlier DAA regimen, achieving high cure rates even in this difficult-to-treat group.
- Simplified treatment pathways — Several countries now allow primary care physicians and even pharmacists (with prescribing authority) to initiate HCV treatment, removing the requirement for specialist gastroenterologists or hepatologists in routine cases.
- Access programs — Generic DAA production under voluntary licensing agreements (particularly for sofosbuvir-based regimens) has dramatically reduced costs in lower-income countries. Generic sofosbuvir/velpatasvir is now available in India, Egypt, and several African nations for under $100 per treatment course.
The WHO’s target is to eliminate hepatitis C as a public health threat by 2030, requiring 90% of diagnosed patients to receive treatment. Current global treatment rates remain far below this goal, primarily due to cost barriers in high-income countries and diagnostic gaps in low-income settings.
Antiviral Medication for Hepatitis B vs Hepatitis C: Key Differences
Many people confuse hepatitis B and hepatitis C treatment — the viruses are biologically distinct, transmitted differently, and respond to completely different antiviral strategies.
Hepatitis C (HCV)
- RNA virus; does not integrate into host DNA
- Curable with 8–12 weeks of DAA therapy
- No vaccine available
- Treatment goal: permanent elimination (SVR)
- Main drugs: sofosbuvir, velpatasvir, glecaprevir, pibrentasvir
Hepatitis B (HBV)
- DNA virus; integrates into host liver cell DNA
- Currently not curable — treatment suppresses replication long-term
- Preventable by vaccine (one of the most effective vaccines available)
- Treatment goal: viral suppression, preventing liver damage
- Main drugs: tenofovir (TDF/TAF), entecavir; pegylated interferon for some
The key distinction: antiviral medication for hepatitis B does not cure the infection in most patients. Drugs like tenofovir disoproxil fumarate (TDF) and entecavir suppress HBV replication to undetectable levels, preventing disease progression — but treatment is typically lifelong. HCV, by contrast, can be eliminated entirely from the body.
This biological difference exists because HBV integrates its genetic material (cccDNA) into the host cell’s nucleus, where current antivirals cannot reach it. HCV remains in the cytoplasm and can be fully eliminated by blocking its replication machinery.
Who Qualifies for Hepatitis C Treatment and What to Expect
Diagnosis and eligibility
Any person with confirmed chronic HCV infection (positive anti-HCV antibody + detectable HCV RNA by PCR) is generally eligible for treatment. Current clinical guidelines from the American Association for the Study of Liver Diseases (AASLD) and the European Association for the Study of the Liver (EASL) recommend treatment for all patients with chronic HCV, regardless of fibrosis stage.
Prior to prescribing, clinicians typically assess: HCV RNA viral load, HCV genotype (though pangenotypic regimens make this less critical), degree of liver fibrosis (via FibroScan or blood tests), kidney function (relevant for sofosbuvir dosing), and potential drug interactions.
What happens during treatment
Most DAA regimens involve one or two pills taken once daily with or without food. The course is 8 or 12 weeks for most patients, extending to 16 weeks only for complex cases (advanced cirrhosis, genotype 3 with prior treatment failure).
Side effects are generally mild compared to interferon-era treatment. The most commonly reported include fatigue, headache, and nausea, particularly in the first two weeks. Drug interactions are the more significant clinical concern — especially with HIV antiretrovirals, certain heart medications, and proton pump inhibitors (which can reduce sofosbuvir absorption if taken simultaneously).
Confirming the cure
At 12 weeks after the final pill, a PCR test for HCV RNA is performed. An undetectable result is defined as SVR12 and is equivalent to a cure. No further antiviral treatment is needed. Follow-up liver function tests are advised periodically, particularly in patients who had advanced fibrosis before treatment, since cirrhosis-related liver cancer risk persists even after viral clearance.
Practical Tips for Anyone Starting Hepatitis C Treatment
- 1Take medication at the same time every day. Consistency maintains stable drug levels in your bloodstream. A missed dose should be taken as soon as remembered on the same day — never double-dose the following day.
- 2Disclose all medications to your prescriber. Drug-drug interactions are the leading cause of DAA treatment failure. This includes over-the-counter medications, herbal supplements (especially St. John’s Wort, which can dramatically reduce DAA blood levels), and antacids.
- 3Avoid alcohol during treatment. Alcohol accelerates liver fibrosis independently of viral load. Even a few drinks per week can work against liver recovery during treatment. Complete abstinence during the 8–12 week course is strongly recommended.
- 4Do not stop treatment early if you feel better. HCV RNA can become undetectable within 2–4 weeks in many patients, but stopping early dramatically increases relapse risk. Complete the full prescribed course regardless of symptom improvement.
- 5Check for access programs before assuming cost is prohibitive. In many countries, DAA therapy is covered under national health programs. In the US, manufacturer patient assistance programs (AbbVie, Gilead) offer free or subsidised medication for uninsured or underinsured patients. Generic options exist in many low- and middle-income countries.
- 6Get tested for hepatitis B co-infection. HBV reactivation has been documented in patients with chronic HBV surface antigen (HBsAg) who undergo HCV treatment. All patients should be tested for HBV before starting DAAs, and those with active HBV may need concurrent HBV suppression therapy.
For official treatment guidelines, refer to the HCV Guidance from AASLD and IDSA — the most comprehensive and regularly updated clinical resource for hepatitis C treatment in the United States. The WHO Hepatitis C Fact Sheet provides global epidemiology data and access program information.
Frequently Asked Questions
Yes. Hepatitis C is curable in 95–99% of patients using direct-acting antiviral drugs. A cure is defined as SVR12 — no detectable HCV RNA in the blood 12 weeks after finishing treatment. This response is durable; relapse occurs in fewer than 1% of patients who achieve SVR12.
Most patients see HCV RNA drop to undetectable levels within 2–4 weeks of starting treatment. However, completing the full 8–12 week course is essential — stopping early significantly increases the risk of viral relapse and potential resistance. Cure is confirmed by a PCR test 12 weeks after the last dose.
The most recently developed regimen is Vosevi (sofosbuvir/velpatasvir/voxilaprevir), a triple-class DAA approved for patients who have previously failed a DAA regimen. For treatment-naive patients, Mavyret (glecaprevir/pibrentasvir) with its 8-week pangenotypic course represents one of the shortest and most effective current options.
Yes — genotype 3 is the most treatment-resistant HCV genotype, particularly in patients with cirrhosis or prior treatment failure. While Epclusa and Mavyret work for treatment-naive genotype 3 patients without cirrhosis, Vosevi for 12 weeks is the preferred option for cirrhotic or previously treated genotype 3 patients, achieving cure rates above 95%.
No. HBV and HCV are completely different viruses requiring different drugs. Hepatitis B is treated with nucleoside/nucleotide analogues like tenofovir or entecavir, which suppress the virus long-term but do not cure it. Hepatitis C is treated with direct-acting antivirals that target HCV-specific proteins and can eliminate the virus entirely in most patients.
Untreated chronic HCV causes progressive liver damage. Over 20 years, approximately 15–30% of untreated patients develop cirrhosis. Cirrhosis carries a 1–5% annual risk of liver cancer. Liver failure and hepatocellular carcinoma from untreated HCV are leading causes of liver transplantation globally. Early treatment prevents virtually all of these outcomes.
Yes. Curing hepatitis C provides no immunity against re-infection. People who continue to engage in behaviours that carry transmission risk — primarily sharing needles or syringes — can contract HCV again after successful treatment. Re-infection is treated the same way as first infection and responds equally well to DAA therapy.
Final Takeaway: Hepatitis C Is Highly Treatable — And Curable
Antiviral medication for hepatitis C has transformed one of the most feared liver diseases into a routinely curable condition. The key facts every diagnosed patient should know:
- Modern DAAs achieve a 95–99% cure rate across all genotypes
- Treatment lasts just 8–12 weeks and is taken as a daily oral pill
- SVR12 (undetectable virus 12 weeks after treatment) equals a permanent cure in over 99% of cases
- Genotype 3 and patients with cirrhosis may need specific regimens — particularly Vosevi for treatment-experienced cases
- Hepatitis B is managed differently — suppression, not cure, is the current treatment goal for HBV
- Drug interactions and HBV co-infection are the main clinical considerations before starting treatment
If you or someone you know has been diagnosed with hepatitis C, the message is clear: treatment works, the side effects are manageable, and the outcome is almost always a cure. The sooner treatment begins, the less cumulative liver damage occurs — and the more liver function can recover after viral clearance.
Speak to a hepatologist or gastroenterologist to confirm your genotype, assess your liver fibrosis stage, and select the right regimen. Cure is within reach for the overwhelming majority of patients.